Class 10 Science Chapter -1 notes
Chapter- 1 Chemical Reactions and Equations
Most of the substance around us undergoes various changes. Based on this Changes are of two types:-
1. Physical Change
2. Chemical Change
Physical Change:-
Some changes are temporary and no new substance is formed in it. These changes are called physical changes.
Chemical changes:-
In some changes the new substance formed in which the reactant or the parent loses its identity to form new substance called product. These changes are permanent changes as we won’t get the reactant back.
• In a chemical change some bonds are broken and some bonds are formed. The products are quite different from the reactants. Therefore it normally can't be reversed.
In which of the following, the identity of initial substance remains unchanged?
(a) Curdling of milk
(b) Formation of crystals by process of crystallisation
(c) Fermentation of grapes
(d) Digestion of food
Answer:
(b): Formation of crystals is a physical change rest others are chemical change.
.
.
Chemical Reaction
Chemical reaction is the process by which two or more substance react with each other to form new substance with different properties.
changes which shows that the chemical reaction has taken place:
(i) Change in state
(ii) Change in colour
(iii) Evolution of gas
(iv) Change in temperature
Chemical Equation
The symbolic representation of a chemical reaction is called chemical equation.
For example:-
Magnesium + oxygen → magnesium oxide
(Reactant) (Product)
•The reactants are written on the left side.
• The products are written on the right side.
Balanced chemical equation
The chemical equation that shows the chemical reaction needs to be balanced.
In balanced chemical equation the number of the atoms in the reactants side is equal to the number of atoms in the products side.
Zn + H2SO4 → ZnSO4 + H2
3Fe (s) + 4H2O (g) → Fe3O4 (s) + 4H2 (g)
• Balancing of a chemical equation is based on the law of conservation of mass
Law of conservation of mass states that mass can neither be created nor destroyed in a chemical reaction. That is, the total mass of the elements present in the products of a chemical reaction has to be equal to the total mass of the elements present in the reactants in a balanced equation.
Balance the following chemical equation:
Na + H3O → NaOH + H2
Hydrogen + Chlorine → Hydrogen chloride
Lead + Copper chloride → Lead chloride + CopperZinc oxide + Carbon → Zinc + Carbon monoxide
ANS -2Na(s) + 2H2O(l) → 2NaOH(aq) + H2(g
H2(g) + Cl2(g) → 2HCl(g)
Pb(s) + CuCl2(aq) → PbCl2(aq)+ Cu(s)
ZnO(s) + C(s) → Zn(s) + CO(g
Types of chemical reaction
Combination reaction: A reaction in which a single product is formed from two or more reactants is known as a combination reaction.
CaO (s) + H2O (l) → Ca(OH)2 (aq)
Calcium oxide reacts vigorously with water to produce slaked lime (calcium hydroxide) releasing a large amount of heat.
• A solution of slaked lime produced by the reaction is used for white washing walls.
• Calcium hydroxide reacts slowly with the carbon dioxide in air to form a thin layer of calcium carbonate on the walls. Calcium carbonate is formed after two to three days of white washing and gives a shiny finish to the walls. It is interesting to note that the chemical formula for marble is also CaCO3.
Ca(OH)2 (aq) + CO2 (g) → CaCO3 (s) + H2O (l)
(slaked lime) (calcium carbonate)
• Burning of coal: C (s) + O2 (g) → CO2 (aq)
• Formation of water: H2 (g) + O2 (g) → H2O (aq)
• Burning of natural gas (Methane): CH4 (g) + O2 (g) → CO2 (g) + H2O (l)
• When magnesium ribbon burns in air, it combines with the oxygen to form magnesium oxide.
2Mg(s) + O2(g) 2MgO(s)
# Study the following equation of a chemical reaction:
H2 + Cl2 → 2HCl
(i) Identify the type of reaction.
(ii) Write a balanced chemical equation of another example of this type of reaction.
Answer:
(i) Combination reaction.
(ii) Another example of combination reaction is![]()
# 1 g of copper powder was taken in a China dish and heated. What change takes place on heating? When hydrogen gas is passed over this heated substance, a visible change is seen in it. Give the chemical equations of reactions, the name and the colour of the products formed in each case.
Answer:
When copper powder is heated in a China dish, the reddish brown surface of copper powder becomes coated with a black substance which is copper oxide.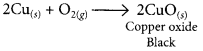
When hydrogen gas is passed over CuO, the black coating on the surface turned reddish brown due to the formation of Cu.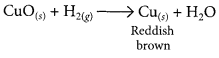
# A compound ‘A’ is used in the manufacture of cement. When dissolved in water, it evolves a large amount of heat and forms compound ‘B’.
(i) Identify A and B.(ii) Write chemical equation for the reaction of A with water.
(iii) List two types of reaction in which this reaction may be classified.
Answer:
(i) A is calcium oxide, CaO which is used in the manufacturing of cement.
B is calcium hydroxide Ca(OH)3.
(iii) The given reaction is a combination reaction.
Example : NH3(g)(g) + HCl(g) → NH4Cl(s)
2NO(g) + 02(g) → 2NO2(g)
Mention with reason the colour changes observe when:
copper powder is strongly heated in the presence of oxygen.
Answer:
When copper powder is strongly heated in presence of oxygen, the reddish brown surface of copper powder becomes coated with a black substance which is copper oxide.
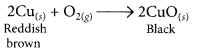
Exothermic reaction:
An exothermic process releases heat, and causes the temperature of the immediate surroundings to rise The rice, potatoes and bread we eat contain carbohydrates. These carbohydrates are broken down to form glucose. This glucose combines with oxygen in the cells of our body and provides energy. The special name of this reaction is respiration is an exothermic reaction.
C6H12O6 (aq) + 6O2 (aq) → 6CO2 + 6H2O (l) + energy
(glucose)
Photosynthesis is a process by which plants utilize carbon dioxide and water in the presence of sunlight to produce glucose and oxygen.![]()
# Calcium oxide reacts vigorously with water to produce slaked lime.
CaO(s) + H2O(l) → Ca(OH)2(aq)
This reaction can be classified as
(A) Combination reaction
(B) Exothermic reaction
(C) Endothermic reaction
(D) Oxidation reaction
Which of the following is a correct option?
(a) (A) and (C)
(b) (C) and (D)
(c) (A), (C) and (D)
(d) (A) and (B)
Answer:
(d) The reaction between CaO and H2O to form Ca(OH)2 is an exothermic combination reaction.
Endothermic reaction:
An endothermic process absorbs heat and cools the surroundings.
The decomposition of vegetable matter into compost is also an example of an endothermic reaction.
.
.
Decomposition reaction:
When single reactant breaks down to give simpler products. This is a decomposition reaction.
Types of Decomposition reaction
• Decomposition reactions which require heat are known as thermolytic
• Decomposition reactions which require light are known as photolytic
• Decomposition reactions which require electricity are known as electrolytic.
White silver chloride turns grey in sunlight. This is due to the decomposition of silver chloride into silver and chlorine by light.
The above reactions are used in black and white photography.
# Identify ‘x’, ‘y’ and ‘z’ in the following reaction :![]()
(a) x = gas; y = reaction condition; z = gas
(b) x = solid; y = liquid; z = gas
(c) x = number of moles of KClO3; y = reaction condition; z = number of molecules of oxygen
(d) x = physical state of KClO3 and KCl;
y = reaction condition, z = physical state of O2.
Answer:![]()
Mention with reason the colour changes observe when:
(i) silver chloride is exposed to sunlight.
Answer:
(i) When white silver chloride is left exposed to sunlight, its colour changes to grey as it decomposes to silver in the presence of sunlight.![]()
This type of reaction is called photodecomposition reaction.
Displacement reaction:
Displacement reaction is a chemical reaction in which a more reactive element displaces a less reactive element from its compound.
Both metals and non-metals take part in displacement reactions.
Reaction of iron nails with copper sulphate solution.
Fe (s) + CuSO4 (aq) → FeSO4 (g) + Cu (s)
Pb (s) + CuCl2 (aq) → PbCl2 (aq) + Cu (s)
An iron knife kept dipped in a blue copper sulphate solution turns the blue solution light green
> As iron is more reactive than copper. So, it displaces Cu from CuSO4 solution and forms ferrous sulphate which is of Light Green Colour. CuSO4 (aq) + Fe (s)→ Fe504 (aq) + Cu(s) Blue colour light green colour
A copper coin is kept in a solution of silver nitrate for some time.
> copper is more reactive than silver, so it will displace silver from its salts olution: Cu(s) + 2AgNO3(aq) Cu(NO3)2(aq)+2Ag(s) So the solution will turn blue due to the formation of copper nitrate
# Mention with reason the colour changes observe when:
a piece of zinc is dropped in copper sulphate solution.
Answer:
When a piece of zinc is dropped in copper sulphate solution, then the blue colour of copper sulphate fades gradually due to the formation of colourless zinc sulphate solution and reddish brown copper metal gets deposited on zinc piece.
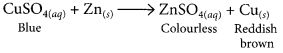
Double Displacement reaction:
A chemical reaction in which there is an exchange of ions between the reactants to give new substances is called double displacement reaction.
Na2(SO)4 (aq) + BaCl2 (aq) → BaSO4 (s) + NaCl (aq)
Precipitation reaction:
An insoluble solid known as precipitate is formed during a doubled displacement reaction. Such reactions are also known as precipitation reactions
For example,
i) When aqueous solution of sodium sulphate is mixed with the aqueous solution or barlum chloride, barium sulphate comes in the form of white precipitate
Na2SO4 (aq)+BaCl2(aq) BaSO(1)+2NaCl(aq)
ii) When aqueous solution of sodium chloride is mixed with the aqueous solution of silver nitrate,silver chloride comes in the form of white precipitate.
# Lead nitrate solution is added to a test tube containing potassium iodide solution.
(a) Write the name and colour of the compound precipitated.
(b) Write the balanced chemical equation for the reaction involved.
(c) Name the type of this reaction justifying your answer. (2020)
Answer:
(a) When lead nitrate is added to potassium iodide then yellow precipitate of lead iodide is formed along with potassium nitrate.
(b) Balanced chemical reaction is as follows :![]()
(c) This type of reaction is called precipitation reaction in which one of the products formed is an insoluble substance or this is also called double displacement reaction.
•lime-water test for the detection of carbon dioxide
>Lime water (calcium hydroxide) combines with carbon dioxide to form a suspension of calcium carbonate which makes lime water milky.
Ca(OH)2(aq) + CO2(g) → CaCO3(s) + H₂0
# When hydrogen sulphide gas is passed through a blue solution of copper sulphate, a black precipitate of copper sulphide is obtained and the sulphuric acid so formed remains in the solution. The reaction is an example of a
(a) combination reaction
(b) displacement reaction
(c) decomposition reaction
(d) double displacement reaction.
Answer:
(d) CuSO4 + H2S → CuS + H2SO4
It is a double displacement reaction as in this reaction CuSO4 and H2S reacting by exchange of Cu2+ and H+ ions to from two new compounds i.e., CuS and H2SO4
# In a double displacement reaction such as the reaction between sodium sulphate solution and barium chloride solution :
(A) exchange of atoms takes place
(B) exchange of ions takes place
(C) a precipitate is produced
(D) an insoluble salt is produced
The correct option is (2020)
(a) (B) and (D)
(b) (A) and (C)
(c) only (B)
(d) (B), (C) and (D)
Answer:
(d) In this reaction exchange of Na+ and Ba2+ ions takes place forming BaSO4 which is a white precipitate i.e., an insoluble salt.
Na2SO4 + BaCl2 → BaSO4 ↓+ 2NaCl
Redox reaction:
A reaction in which oxidation and reduction take place simultaneously in a reaction, is known as a redox reaction.
Oxidation: This process involves gain of oxygen or loss of hydrogen.
Reduction: This process involves gain of hydrogen or loss of oxygen.
Oxidizing Agent
It is the substance which gives oxygen or gains hydrogen.
Or it the substance which is reduced itself and oxidizes other.
Reducing Agent
It is the substance which gives hydrogen or gains oxygen.
Or it the substance which is oxidized itself and reduces other.
Oxidation is the process which involves loss of electrons but reduction is the process which involves gain of electrons.
State the type of chemical reactions, represented by the following equations :
(a) A + BC → AC + B
(b) A + B → C
(c) PQ + RS → PS + RQ
(d) A2O3 + 2B → B2O3 + 2A
Answer:
(a) Displacement reaction.
(b) Combination reaction.
(c) Double displacement reaction.
(d) Displacement reaction or redox reaction.
.
.
Corrosion
The process of slow conversion of metals into their undesirable compounds due to their reaction with oxygen, water, acids, gases etc. present in the atmosphere is called corrosion.
Rusting – Iron when reacts with oxygen and moisture forms red substance called rust.
Rust is hydrated ferric oxide (Fe2O3.xH2O)
Conditions necessary for rusting:
i) Open surfaces of the metal.
ii) Presence of air (Oxygen).
iii) Presence of moisture (water).
• Rusting of iron cannot take place in distilled water because it neither contains dissolved oxygen nor CO₂ both are essential for rusting of iron.
Harmful Effect of Rusting:
Hydrated iron (II) oxide is brittle substance and moves away from the surface thus the object is damaged. The objects get holes, cavities and rough surface. Conditions necessary for rusting
Advantages of corrosion:
Aluminium when on exposed to air, gets coated with a protective layer of aluminium oxide. This protects the metal underneath from further corrosion and damage.
The marble statues often slowly get corroded when kept in open for a long time.
>SO₂ ,NO₂ gases are released into the atmosphere from various sources. These dissolve in rain water to give acid which corrodes marble statue
2S02 +02 →2S03: H₂0+S03 →H₂S04
Rancidity
The taste and odour of food materials containing fat and oil changes when they are left exposed to air for long time due to which the food become stale and gives a bad taste or smell. This is called rancidity.
It is caused due to oxidation of fat and oil present in food material.
Prevention:-
i) Keeping the food at low temperature
ii) Keeping food item in air tight containers
iii) By filling nitrogen in the food storage bags
iv) by adding antioxidants to the food materials.
.
.